Coordination Compounds chapter में एक concept ऐसा है जिसे समझते ही पूरा chapter आसान लगने लगता है — और वह है Coordination Number (CN)।
अगर आपने यह topic clear कर लिया, तो आप आसानी से समझ पाएंगे:
✅ Geometry
✅ Hybridization
✅ Bonding
✅ Isomerism
✅ Nomenclature
Teacher Truth ⭐
👉 “Coordination Number is the backbone of Coordination Chemistry.”
आज हम इसे zero level से advanced तक समझेंगे — बिल्कुल classroom teaching style में।
Online Ways to Make Money from Home घर बैठे पैसे कमाने के बेहतरीन तरीके
How I Made My First ₹10,000 Online – Step-by-Step Guide
🔥 Coordination Number क्या होता है?
✅ Definition (Exam Ready)
Coordination Number = Number of donor atoms directly bonded to the central metal atom/ion.
👉 Hindi में:
समन्वय संख्या वह संख्या है जो बताती है कि कितने donor atoms सीधे central metal से जुड़े हैं।
🧠 Easy Language में समझें:
Imagine metal is a celebrity 😄
और ligands उसके आसपास fans की तरह खड़े हैं।
👉 Fans की total संख्या = Coordination Number.
Simple!
⚡ Coordination Compound Recap
Example:
👉 [Co(NH₃)₆]³⁺
✔ Central metal → Co
✔ Ligands → NH₃
NH₃ के nitrogen donor atom से bond बनता है।
👉 Total donors = 6
⭐ Therefore → Coordination Number = 6
🌟 Why Coordination Number is Important?
Without knowing CN…
❌ Geometry नहीं पता चलेगी
❌ Hybridization guess नहीं कर पाएंगे
❌ Magnetic behavior unclear रहेगा
❌ Isomerism समझ नहीं आएगा
👉 CN decides the shape and structure of the complex.
Teacher Tip ⭐
“Find CN first — everything else becomes easy.”
🎯 How to Calculate Coordination Number?
Students अक्सर confuse हो जाते हैं — लेकिन यह बेहद आसान है।
👉 बस donor atoms count करो!
Example 1:
[Cu(NH₃)₄]²⁺
NH₃ → 1 donor each
Total = 4
👉 CN = 4
Example 2:
[Co(en)₃]³⁺
en (ethylenediamine) is bidentate.
1 en → 2 donor atoms
3 en → 6 donors
👉 CN = 6
⭐ IMPORTANT:
Ligands count मत करो — donor atoms count करो!
🧬 Coordination Number vs Oxidation State
Students अक्सर दोनों mix कर देते हैं.
| Feature | Coordination Number | Oxidation State |
| Meaning | Donor atoms | Charge on metal |
| Depends on | Ligands | Electron loss |
| Always positive? | Yes | Can vary |
Example:
[Fe(CN)₆]⁴⁻
✔ CN = 6
✔ Oxidation state = +2
Different concepts!
🔥 Coordination Number and Denticity
This is VERY important for boards 🚨
👉 Denticity = donor atoms per ligand.
Formula:
⭐ CN = Denticity × Number of ligands
Example:
Ligand = EDTA
Denticity = 6
Only ONE EDTA needed.
👉 CN = 6
Amazing, right? 🙂
🌈 Most Common Coordination Numbers
Exam में बार-बार पूछे जाते हैं।
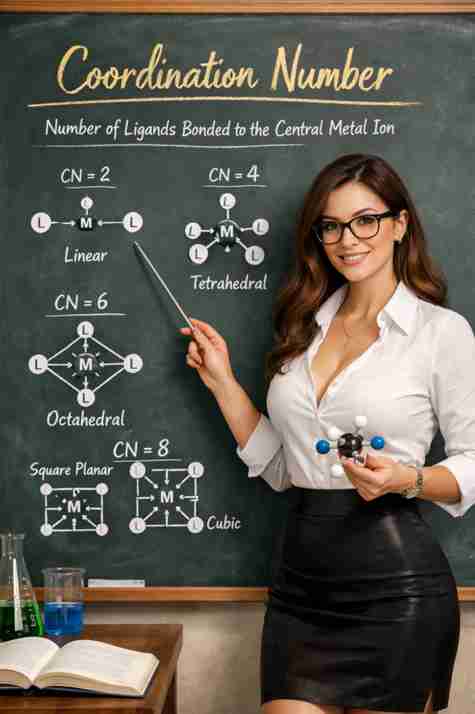
✅ Coordination Number = 2
Geometry → Linear
Bond angle → 180°
Example:
👉 [Ag(NH₃)₂]⁺
Shape is straight line.
✅ Coordination Number = 4
Two possibilities:
✔ Tetrahedral
✔ Square Planar
Tetrahedral:
Bond angle → 109.5°
Example:
👉 [NiCl₄]²⁻
Usually weak ligands → tetrahedral.
Square Planar:
Bond angle → 90°
Example:
👉 [Pt(NH₃)₂Cl₂]
Common in d⁸ metals like Pt²⁺.
Teacher Trick ⭐
“Pt loves square planar.”
✅ Coordination Number = 6
Most common!
Geometry → Octahedral
Bond angle → 90°
Example:
👉 [Co(NH₃)₆]³⁺
Very stable structure.
✅ Coordination Number = 8
Rare but possible.
Geometry → Square antiprismatic.
Example:
👉 [Mo(CN)₈]⁴⁻
Advanced level 🙂
⚙ Coordination Number and Hybridization
CN helps predict hybridization.
| CN | Hybridization | Geometry |
| 2 | sp | Linear |
| 4 | sp³ / dsp² | Tetrahedral / Square planar |
| 6 | d²sp³ / sp³d² | Octahedral |
Exam Favorite ⭐
🧪 Factors Affecting Coordination Number
Great conceptual question!
1️⃣ Size of Metal Ion
Large metal → more ligands fit.
👉 CN increases.
2️⃣ Size of Ligands
Bulky ligands → less space.
👉 CN decreases.
3️⃣ Charge on Metal
Higher charge → stronger attraction.
👉 More ligands attach.
4️⃣ Steric Hindrance
Crowding reduces CN.
🔗 Chelation and Coordination Number
Polydentate ligands increase CN quickly.
Example:
3 bidentate ligands → CN = 6
Chelate complexes are more stable.
🎨 Coordination Number and Color
CN affects d-orbital splitting.
Different splitting → different color.
Example:
Ni²⁺ tetrahedral → blue
Ni²⁺ octahedral → green
Chemistry is colorful 😄
🧲 Coordination Number and Magnetism
Geometry changes electron pairing.
👉 Tetrahedral → often paramagnetic
👉 Square planar → often diamagnetic
Board loves this concept!
⚠ Common Mistakes Students Make
❌ Counting ligands instead of donors
❌ Confusing CN with oxidation state
❌ Forgetting denticity
❌ Assuming geometry without CN
Avoid these → Easy marks.
🧠 Memory Tricks (Topper Secrets)
⭐ CN = “Count Neighbors”
⭐ Octahedral → Think Octopus (8 arms around 😄)
(Actually 6 bonds — but helps memory!)
🏆 Board Exam Strategy
📚 ऐसे ही exam-focused notes के लिए visit करें:
👉 gurugyanam.online
Want guaranteed marks?
Focus on:
✅ Definition
✅ Calculation
✅ Geometry
✅ Denticity relation
✅ Hybridization
Practice daily!
🧪 Solved Board-Level Questions
Question:
Find CN in [Cr(H₂O)₄Cl₂]⁺
H₂O → 4 donors
Cl⁻ → 2 donors
Total = 6
👉 CN = 6
Question:
CN in [Ni(en)₂Cl₂]
en → bidentate → 4 donors
Cl → 2 donors
👉 CN = 6
🌍 Real-Life Importance
Coordination number helps design:
✅ Cancer drugs
✅ Catalysts
✅ Metal extraction processes
✅ Biological molecules
Example:
Hemoglobin CN = 6
Life depends on coordination chemistry!
⭐ Most Expected Board Questions
👉 Define coordination number.
👉 How to calculate CN?
👉 CN vs oxidation state.
👉 CN and geometry.
👉 Effect of denticity.
Prepare these → Score high.
❓ Top 20 FAQs – Coordination Number (समन्वय संख्या)
Q1. समन्वय संख्या क्या है?
Ans. Number of donor atoms bonded to metal.
Q2. CN कैसे find करें?
Ans. Donor atoms count करें।
Q3. CN = ligands?
Ans. Not always!
Q4. CN vs oxidation state same है?
Ans. No.
Q5. Most common CN?
- Ans.
Q6. CN = 2 geometry?
Ans. Linear.
Q7. CN = 4 geometry?
Ans. Tetrahedral / Square planar.
Q8. CN = 6 geometry?
Ans. Octahedral.
Q9. Bidentate ligand denticity?
- Ans.
Q10. EDTA denticity?
- Ans.
Q11. CN always integer?
Ans. Yes.
Q12. Can CN be 1?
Ans. Rare.
Q13. Large metal → CN?
Ans. Higher.
Q14. Bulky ligand → CN?
Ans. Lower.
Q15. CN affects color?
Ans. Yes!
Q16. CN affects magnetism?
Ans. Yes.
Q17. CN formula?
Ans. Donors count.
Q18. Most stable CN?
Ans. Often 6.
Q19. Board exam important?
Ans. VERY HIGH.
Q20. Trick to master
Ans. Practice examples.
🏁 Final Teacher Advice
समन्वय संख्या डरने वाला topic नहीं है — यह logic based है।
👉 पहले donor atoms identify करें
👉 फिर geometry सोचें
👉 फिर hybridization
Remember:
⭐ “Understand structure — and chemistry becomes easy.”
Daily 15 minutes revise करें — और यह chapter आपका strongest बन जाएगा।
📚 ऐसे ही exam-focused notes के लिए visit करें:
👉 gurugyanam.online
Study smart. Stay confident. Crack your boards. 🚀